By Michael CasoBackground and Objectives
The rationale and benefits of having a scientific platform and subsequent publication plan for a new drug entity are well established in the biopharmaceutical industry. The ability to disseminate clinical concepts and research results to as wide an audience as possible has demonstrated significant value to biopharmaceutical organizations. It is true that we have successfully developed a comprehensive set of strategies and tactics designed to identify market gaps and key scientific communications, as well as research appropriate publication and presentation venues. However, simply planning a publication placement does not maximize the value of that article. Publishing the scientific data that our preclinical research and clinical development teams have worked hard to produce validates or disproves the original scientific hypothesis. So why do we think that simply publishing the content is all that needs to happen? Biopharmaceutical content dissemination needs to catch up with the approaches taken in other industries, proactively expanding the audience and digitally presenting key messages to reach a larger range of key targets through currently available electronic communication vehicles. So yes, while the terms “marketing” and publication planning are not normally used in the same sentence, there are ways to accomplish a myriad of product objectives at pre-publication, publication launch and post-publication timeframes. At Omni Healthcare Communications (Omni-HC) we believe all publications should:
Concept Overview As stated previously, the development of a product’s scientific platform forms the foundation upon which all communications activities can be developed. Key to the early awareness and long-term success of a product are the:
Scientific Platform Stakeholder Expansion The mission of a truly comprehensive scientific platform should be to Educate, Empower, and Engage all key stakeholders, including providers, payers and patients. On the other hand, Medical Affairs teams don’t have as strong a relationship with all stakeholders that allows the ability to publish data once and all will see it, especially if published in a journal behind a pay access wall. In the digital age, traditional publications and the underlying communication plan can be enhanced, not just by more publications, but also by using electronic means to enhance and disseminate key information. Key Issues to Address
Key Opportunities for Action Pre-publication communications Creating awareness of upcoming publications is a productive way to prepare targeted audiences for your soon-to-be-published manuscript. Many journals are now providing services designed to alert subscriber targeted audiences to upcoming publications. Specific opportunities are outlined on publisher websites and should be considered into the publication plan. Enhancing awareness at publication The use of social media tactics commonly used in reaching consumer audiences has now reached the medical communications industry. A well-developed plan of action for a publication plan would include the following:
Post-publication follow-through In the current medical communications environment, we are all being required to demonstrate value for the services we provide. This is also the case for publication planning. Simply submitting a publication to a journal is not sufficient. What are potential opportunities to capitalize on manuscripts in print, as well as show an impact of our publication planning recommendations?
Scientific publications have been the archive of scientific evidence for over a century. Within the scientific archive, it is estimated there are almost 50 million scientific papers have been published and the number of papers published each year is increasing. Although simply publishing the data gives a reference point for future use, much like anything else, the more stakeholders are exposed to the data and message the better the scientific exchange. All these areas mentioned above help to broaden the audience for the scientific communication and increase the value of the effort to the company. At Omni-HC, we actively consider all the various ways that publications and other communications may be enhanced to generate the maximum value from the company effort. We strive to not only efficiently develop communications for the various key audiences, but also to expand the audience for that communication and message.
0 Comments
By Michael CasoBiopharmaceutical companies pride themselves on conducting comprehensive research activities. These research initiatives could be clinical programs to determine the viability of a new chemical entity to satisfy an unmet medical need, market access programs, or more recently, patient-centric needs of targeted populations. Why then have these same companies virtually ignored the medical, social and cultural needs of the fastest growing segment of the U.S. population?
Ethnic and racial minorities currently comprise over 30 percent of the U.S. population and will grow to 50 percent by 2050. In fact, in many geographical areas, minority populations represent an “emergent majority” — a collective majority of minorities. Additionally, this population can have unique responses to medications and are differentially impacted by a number of common disease states for which they are diagnosed later and have poorer control or survival rates than majority populations. It would appear that there exists here a nexus of needs and opportunities that should appeal to the biopharmaceutical industry, where a focus on a medical need can result in a healthier population and a healthier bottom line. This is the first of a three-part posting that will provide supportive data that identifies the need, demonstrates targeting opportunities and provides specific strategies and tactics designed to positively impact complementary health disparity reduction and business goals. Opportunities for Targeting: Disease States and Geography Disease States Disparities in healthcare are evident in the diagnosis and treatment of specific health conditions, in the utilization of preventive services and in health outcomes. These disparities may be caused by unique reactions to medicines, cultural or social factors. In fact, the majority of published research indicates that minorities are less likely than whites to receive needed services. In Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, the Institute of Medicine (IOM) of the National Academies, reports: African-Americans and Hispanics tend to receive a lower quality of healthcare across a range of disease areas; disparities are found across a range of clinical settings; and the disparities in care are associated with higher mortality among minorities. Any institution that impacts public health — academia, medicine and social services, etc. — has a role to play in reducing disparities in healthcare; pharmaceutical companies are no exception. It would be challenging to find a pharmaceutical company without a product in at least one of the core six disease states identified by the U.S. Department of Health and Human Services (HHS) Office of Minority Health (OHM), in which racial and ethnic minorities experience serious disparities in health access and outcomes, as well as other diseases which differentially impact minority populations:
In addition, the following diseases and conditions disproportionately impact racial and ethnic minorities: Disease Disparities Mental health
While providing a tremendous service by addressing critical unmet medical needs, pharmaceutical companies will expand business opportunities by reaching minority populations. Analyzing and communicating data about these markets can improve pharmaceutical company efforts in improving outcomes and delivering assistance to the healthcare system through improved population management. The improved outcomes and population health management assistance will increase product demand, enhance relationships with growing physician and patient populations, and lead to networking opportunities within governmental health agencies, ethnic medical associations and community-based organizations. Geography While a focus on specific disease states is critical, so too is the ability to impact specific population bases. In this manner, it is more likely that limited resources can have a significant and measurable impact that can then be duplicated in other areas of need. Historically, significant minority population bases exist in major metropolitan areas. Wikipedia, as well as government census reports provide listings of cities for black, Hispanic and Asian populations and these tables can be used for creating an initiative for a single population. However, if your goal is to have a comprehensive initiative across all three of these minority groups, a comparative analysis of metropolitan areas would be required. A suggested list of targeted cities for initiatives impacting black, Hispanic, and Asian populations would include the following:
Developing a Plan for Engagement and Collaboration Now that we have addressed the why, what and where, let’s review options for how. The development of any plan for engaging minority physicians and patients should exhibit a number of key characteristics:
Proposed Strategies and Tactics Having provided the characteristics listed above, the following are suggested strategies along with associated tactics:
As the U.S. population demographics shift, so too must the focus of the pharmaceutical industry. As stated previously in this publication, there exists here a nexus of needs and opportunities with minority populations, where a focus on a medical need can result in a healthier population and a healthier bottom line. By Michael CasoOpportunities for Targeting – Disease States and Geography
This is the second of a 3-part series that is geared to provide supportive data that identifies the need, demonstrates targeting opportunities, and provides specific strategies and tactics designed to positively impact complementary health disparity reduction and business goals. Disease States Disparities in healthcare are evident in the diagnosis and treatment of specific health conditions, in the utilization of preventive services, and in health outcomes. These disparities may be caused by unique reactions to medicines, cultural or social factors. In fact, the majority of published research indicates that minorities are less likely than whites to receive needed services. In Un-equal Treatment: Confronting Racial and Ethnic Disparities in Healthcare, the Institute of Medicine (IOM), reports: African-Americans and Hispanics tend to receive a lower quality of healthcare across a range of disease areas; disparities are found across a range of clinical settings; and disparities in care are associated with higher mortality among minorities. Any institution that impacts public health — academia, medicine and social services, etc. — has a role to play in reducing disparities in healthcare; pharmaceutical companies are no exception. It would be challenging to find a pharmaceutical company without a product in at least one of the core six disease states identified by HHS, Office of Minority Health, in which racial and ethnic minorities experience serious disparities in health access and outcomes, as well as other diseases which differentially impact minority populations:
Mental health Disparities
Hepatitis Disparities
Syphilis Disparities
Tuberculosis (TB) Disparities
Lupus Disparities
Asthma Disparities
Stroke Disparities
While providing a tremendous service by addressing critical unmet medical needs, pharmaceutical companies will expand business opportunities by reaching minority populations. Analyzing and communicating data about these markets can improve pharmaceutical company efforts in improving outcomes and delivering assistance to the healthcare system through improved population management. The improved outcomes and population health management assistance will increase product demand, enhance relationships with growing physician and patient populations, and lead to networking opportunities within governmental health agencies, ethnic medical associations and community-based organizations. Geography While a focus on specific disease states is critical, so too is the ability to impact specific population bases. In this manner, it is more likely that limited resources can have a significant and measurable impact that can then be duplicated in other areas of need. Historically, significant minority population bases exist in major metropolitan areas. Wikipedia, as well as government census reports provide listings of cities for African-American, Hispanic and Asian populations and these tables can be used for creating an initiative for a single population. However, if your goal is to have a comprehensive initiative across all three of these minority groups, a comparative analysis of metropolitan areas would be required. A suggested list of targeted cities for initiatives impacting African-American, Hispanic and Asian populations would include the following:
By Steve Casey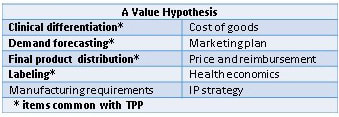 Early-stage companies commonly focus solely on investors and regulatory stakeholders and overlook other stakeholders who are critical in optimizing the later development-commercialization pathway. Addressing these other stakeholders maximizes the “downstream” value of any product. Such stakeholders are gatekeepers critical to life-cycle areas such as market access, reimbursement, and medical education. Even investors in early-stage companies are now considering the impact the “downstream” stakeholders have on their ability to secure financial exits. Today’s newly minted healthcare-product companies are frequently being asked to demonstrate both regulatory viability and commercial potential at the earliest stages. Long gone are the days when a product would “sell itself” and companies could count on guaranteed reimbursement and rapt attention from healthcare providers. Rather, today’s stakeholders are focused on specific therapies with defined risks and demonstrated healthcare benefit. As a result of these changes, a broader definition of product success must be established during early development to generate a rational basis and plan of action for both product approval and commercial success.
Engineer the Product Value It has long been understood that an evaluation of commercial potential during the early stages of clinical development in parallel with a well-developed regulatory strategy can greatly increase the value of a product. The integration of market forecasting, health economics, reimbursement and anticipated medical practice input leads to a development strategy that engineers more value and is more cohesive than a traditional staged approach. While commonplace in large pharmaceutical companies, this integration can be difficult for smaller companies where resources are limited, information is incomplete, and, frankly, product commercialization is viewed as the job of whatever company decides to license the product. While many legitimate limitations exist in the ability to describe a product’s potential completely at an early-development stage, future success will be determined by how well companies are able to structure integrated development programs to meet the needs of all stakeholders. How to Get There from Here Fortunately, it doesn’t require armies of healthcare professionals, scientists, and MBAs to develop an integrated clinical, regulatory, and commercial development plan. If a company takes a structured approach to establishing the program goals, much of the information is common to all perspectives. It has been said that the value of a product is driven by its product label; this is true if the commercial aspects of a healthcare product’s business are limited to communicating only what is in the label. To help guide development programs the FDA has provided a good starting point for drug developers with the Target Product Profile (TPP). The FDA TPP template follows the major categories of a package insert, allowing developers to forecast the desired target product attributes and track progress against this profile. The TPP tool is well understood and should always be used by companies developing new therapies. However, the TPP is focused primarily on regulatory approval and is not sufficient to describe fully the commercial attributes of a successful therapeutic. In parallel to the development of the TPP, companies should also consider creating a value hypothesis. The Value Hypothesis A value hypothesis considers the desired global commercial attributes for the therapeutic. As with the TPP, for early-stage programs the value hypothesis will be more general and aspirational in nature, but the real value of the hypothesis for an early-stage development program is in its creation. The development of the hypothesis, the identification of unknowns, and the specific questions that are generated as a result of the analysis, will all guide where early-stage candidates may warrant additional commercialization diligence. While all items will be important eventually, prioritizing where a company should focus limited resources in the value hypothesis is an important decision. A Value Hypothesis The importance of intellectual property (IP) protection in the biopharma industry dictates that IP strategy should be the top priority. Although many companies have competent legal staff handling their IP strategy, many early-stage programs stop with IP and provide limited focus to the other important elements that define commercial value of an early-stage therapeutic (ie. clinical differentiation, pricing/reimbursement, and demand forecasting). An exhaustive analysis of these commercial components is not practical or necessary at the early stages of product development; however, any company seeking to invest $10M or more in getting to a Phase 1 clinical study should have a clear understanding of the commercial assumptions for their product. But a value-hypothesis analysis is extremely valuable and better informs clinical decisions. Starting with a value hypothesis, therapeutic developers will improve clarity of the development/commercialization pathway, enhance confidence in the clinical program, and create a solid understanding to address stakeholder needs. This integration of regulatory and commercial factors will be critically important for product success in the new healthcare-market dynamics. The senior staff at Omni Healthcare Communications have extensive expertise in creating value hypotheses for developmental agents during their experiences while working at pharma companies and as proactive agency partners. Omni welcomes the opportunity to bring an unparalleled depth and breadth of experience to maximize the benefit and value that your product can achieve. by Michael Caso  Biopharmaceutical companies pride themselves on conducting comprehensive research activities. These could be clinical programs to determine the viability of a new chemical entity to satisfy an unmet medical need, market access programs, or more recently, patient-centric needs of targeted populations. Why then have these same companies virtually ignored the medical, social and cultural needs of the fastest growing segment of the U.S. population? Ethnic and racial minorities currently comprise over 30 percent of the U.S. population and will grow to 50 percent by 2050. In fact, in many geographical areas, minority populations represent an “emergent majority” — a collective majority of minorities. Additionally, this population can have unique responses to medications and are differentially impacted by a number of common disease states for which they are diagnosed later and have poorer control or survival rates than majority populations. It would appear that there exists a nexus of needs and opportunities that should appeal to the biopharmaceutical industry, where a focus on a medical need can result in a healthier population and a healthier bottom line. This is the first of a three-part posting that will provide supportive data that identifies the need, demonstrates targeting opportunities, and provides specific strategies and tactics designed to positively impact complementary health disparity reduction and business goals. By Kevin D. Pawley “Market Access is a dynamic process for building an ever-growing, ever-changing value story that validates a product’s price/position in healthcare practice”—at least, that’s what I claimed in my last article. Trying to see into the future, I’ve been reading, searching, studying, and talking with people to define the term and predict where this area of the healthcare industry, Market Access, is heading. The irony is that Market Access is evolving so rapidly that I settled on a definition that was equally changeable…my definition is more vague than I wanted it to be: it includes three terms—dynamic, ever-growing, ever-changing—that guarantee constant change. But I believe in the foundation of my definition: “Market Access is a process for building a value story.” Logically, the next question is, “What are the building blocks of that value story…how do we construct the story?” I have found answers as simple as “In the US, just build your value story with the Academy of Managed Care Pharmacy’s (AMCP) dossier template.” Or as complex as “Building a Market Access value story is finding the nexus of disease, product, economics, models and markets.” The answer must lie somewhere in between those two: using a “template” and “finding a nexus.” I’ve arrived at an answer to the question—but because of the ever-changing nature of Market Access, I am open to everyone’s input to correct my answer or expand on it, but please don’t just dismiss it! Clearly, Market Access positioning and the value story to support a product in the US begins with the AMCP Format for Formulary Submissions (now in version 4.0)…but I offer the following “high altitude” view in answer to the question, “What are the building blocks of that value story?” The Four “E”s of Market Access Epidemiology: The value of any product begins with a comprehensive understanding of the disease: pathophysiology and presentation; risk factors; diagnostic pathways; scope and impact in the target population; practical impact on healthcare systems; and current clinical options. Epidemiology considerations should include real-world questions of “concomitant conditions” that may come into play…who are the subpopulations commonly found in this group—additional diseases or conditions (renal, hepatic, cardiovascular, etc.) that impact the patient, treatment decisions, or healthcare system capabilities. Ultimately, the epidemiology information needed for inclusion in the value story may be modest compared with the clinical and economic data, but it creates a framework for all the other evidence: who has the disease? How does it affect their lives, their jobs, the people around them? How does it affect the healthcare system they belong to? Efficacy: We can presume that any product for which a value story is being built must have demonstrated its efficacy. But efficacy is a broad topic of consideration: clinical efficacy and effectiveness, safety and tolerability, comparative effectiveness versus standard treatment (where appropriate), and new levels/aspects of efficacy compared to currently available treatments. Importantly, the value of efficacy may be relative—does the product offer a unique aspect to its efficacy? Best-in-class efficacy in a high-risk specific subpopulation? Can its efficacy (ie, responders) be predicted based on genetic or clinical markers? The AMCP Format for Formulary Submissions discusses heterogeneity of treatment effect—"Individual patient variability, variability within populations studied, and variability between clinical studies”—that may be a special consideration in determining the value of a product…perhaps filling a specific unmet need in seemingly refractory patients. Economics: The economic impact of any product is also an ever-changing issue. When a product first enters the market, its economic status is based on modeling, which itself is based on subjective assumptions and modifiable variables. In an economic model, change a price or an incidence of disease or predicted clinical events and the economic picture will change greatly. The solution has been to create several coordinated models—clinical events, life expectancy, and quality-adjusted life-years (QALYs)—to create reliably interpretable scenarios. Over time, the assumptions included in the models give way to or are refined by real-world evidence, moving the data away from subjectivity and closer to reality. Ultimately, the economic value of a product will depend on patient, provider, healthcare-system, disease-state, and treatment variables. Everything Else: For want of a better term, this last category encompasses all of the other potential factors that feed into a “value” proposition. Everything and anything may impact the value story depending on the real-world experience of having, diagnosing, treating, and covering a disease.
Which brings me to my summation: the Four “E”s of Market Access can be summed up with a single E: Evidence. In days gone by, Sales & Marketing of a pharmaceutical product was a matter of “features and benefits.” Today, creating a space for a pharmaceutical product—Market Access—is a matter of “features and evidence.” To tell the ever-changing value story that creates a product’s Market Access, the new world requires that Epidemiology, Efficacy, Economics, and Everything Else be supported by Evidence that proves the case. By Dana Randall, PharmD and Kevin D. Pawley  Academic scientists share a mantra: “Publish or perish.” The cornerstone of the scientific process—and the accurate record of it—is peer-review publication of studies in scientific journals. Without publications in such reputable journals, flawed studies and their results may gain impact and real scientific advances may be lost. The continual practice of holding scientific developments up to peer review and documenting by publication the scientific process from concept through completion guarantees those continual developments. Similarly, publications in reputable peer-review medical journals guarantee continual development in the world of medicine. But in the world of medicine, lives may be at stake…making the rapid publication of important medical developments vital. Today’s highly regulated world creates challenges to rapid publication, but my tenet remains, Physicians like data, but they don’t like “data on file.” Thus, the challenge for pharmaceutical companies is to engage the peer-review and publication process efficiently or risk significant delays and roadblocks in a product’s reaching its full benefit and potential. To overcome that challenge—to engage the process efficiently—pharmaceutical companies regularly develop publication plans: planning and disseminating scientific and clinical data on a drug to healthcare professionals (HCPs) by presentation at medical congress/society meetings and publication in peer-review journals. Such planning accounts for a number of variables affecting:
1. Benefit to your product by maximizing the reach and clarity of your clinical data. Plainly stated, clinical data define a product—its use, dosage, benefits, risks, appropriate patient selection…it is the heart and soul of a product. It underwrites the new drug application; it defines clinical use; it creates perceived value. Thus how the drug is introduced, used, and perceived will be based (certainly at launch) on the quality, scope, and scale of the published clinical data. It falls to the owners of the data to create a plan for comprehensive and cohesive disclosure of data to those who need it most—prescribing clinicians. But publication also satisfies legal, ethical, and industry obligations:
2. Benefit to the market by placing your product “in context” within the current therapeutic area. Development of a publication plan is a process of thorough analysis of existing literature about the full therapeutic area, the disease and its diagnosis, as well as treatment options and practices. Additionally, an important element of the process is a “gap” analysis—finding things that aren’t yet well-defined, finding gaps where new clinical data or expanded expert opinion may enhance clinical understanding and practice. If a product is first-in-class, a comprehensive publication plan is particularly important, for example, to introduce a new mechanism of action or define where the drug fits in the clinical picture: Will it be used as a first-line agent? Second-line agent? Concomitant with other drugs? After other therapies fail? Publication of new clinical data expands the existing literature and expands the foundation of evidence-based decision-making. Timely publication of clinical data positions your product to be included in new or updated treatment guidelines. Planned publications that account for the full context of a therapeutic area—diagnosis and treatment—can help place your product in a clearly defined position at launch. 3. Benefit to prescribing HCPs by fostering consistent delivery of scientific communication points across all media. By planning and carefully executing publications, a consistent scientific foundation is established to support the product—consistent language describing mechanism of action, product features, and clinical efficacy and safety. This published body of evidence supports all future product communications, from press releases to promotional materials to patient- and HCP-education materials.  Figure 2. Key stakeholders in the drug development and publication planning processes. Figure 2. Key stakeholders in the drug development and publication planning processes. 4. Benefit to all members of the product development team with a systematic dissemination of all data. A publication plan provides a roadmap and systematic approach to data dissemination for all product stakeholders—drug discovery, clinical development, health economics and outcomes research (HEOR), regulatory affairs. Naturally, each aspect of drug development contributes to the product’s comprehensive profile and clinical use. Publication planning keeps everyone aware, coordinated, and consistent throughout the life of the product. 5. Benefit to all members of the treatment team by delivering key data to key audiences…who needs to hear what and when. A publication plan should address the varying information needs of HCPs across the spectrum of care—specialist and general physicians, nurses, nurse practitioners, physician assistants. Different audiences may need to understand preclinical data versus clinical trial data versus HEOR data. For products with multiple indications, the publication plan should target multiple specialties or multiple types of prescribers; for other products, specialists may be the chief prescribers and thus the main audience; but primary care physicians and allied health professionals need appropriate information, too. 6. Benefit to regulatory bodies and payors by providing clinical context for safety and efficacy, establishing evidence-based value for a product. In a single peer-reviewed publication, authors provide a clinical context and a rational for the trial, and the clinical significance of results. But with today’s escalating healthcare costs and growing demands to control them, regulatory bodies and payors compile data across published studies to establish an evidence-based “value” of a product (ie, AMCP Dossier, Global Value Dossier). A publication plan coordinated across clinical trials ensures a peer-reviewed source for establishing a product’s value in the context of existing medical literature and usual clinical practice. 7. Benefit to the patient by clarifying and addressing all issues that may affect acceptance and appropriate use—even optimal use—of the product. Clinical trial-based data form the core of a publication plan, but additional types of publications may enhance acceptance of your product and directly benefit patients’ understanding of their therapy, ie, a publication describing how an injectable’s unique delivery system improves medication adherence compared with existing delivery systems. HEOR data are increasingly important for acceptance of a new agent in a crowded market, ie, a cost-effectiveness analysis that demonstrates a lower annual cost of your product compared with a well-established competitor or long-term outcomes data demonstrating a reduction in hospitalizations or improved quality of life among patients receiving your product versus a competitor. After a product has been on the market, feedback from the field (eg, medical science liaisons, sales representatives) may inform additional analyses and publications that could provide prescribers with additional patient-centric data. 8. Benefit to the world of medical science by advancing and enriching peer-reviewed medical literature. The peer-reviewed publication process serves a medical/scientific need to validate the data within the medical/scientific community. The medical literature included in PubMed (the US National Library of Medicine and National Institutes of Health online library), includes more than 27 million studies—a phenomenal collection of the progress of mankind’s knowledge and understanding. Where once many illnesses had no name, now they are well-defined and treatable, even curable. Where once disease processes and treatment mechanisms were unknown, now they are clearly understood. For example, over the decades the discussion of autoimmune diseases has moved from inflammation to immune response, from anti-inflammatories to immunomodulation and biologics. Effective publication planning creates a path for advancing and enriching our knowledge. Publications and the publication plan are the core of a pharmaceutical product’s entry to the market and to its acceptance in clinical practice. Thus, publication planning should begin early in the product life cycle, anticipate change, and be adaptable as additional data become available and as the market evolves…because the goal is to provide benefits across the spectrum. By Kevin D. Pawley  I’ve enjoyed a long career in medical communications, a business that has changed as constantly and quickly as the pharma market that it supports. Among all the constant change has been the rise, expansion, and confusion of a thing called “Market Access.” A long time ago, Market Access was only a matter of “negotiating a price with payers.” As payers began to manage their costs more aggressively—based on utilization data that piled into their databases—the challenges of offering the “right price” grew more complex. Outcomes became an important addition to clinical efficacy; economic impact became an important addition as healthcare costs climbed; now, real-world evidence has provided a powerful perspective as the volume of utilization data has grown to astronomical size and modeling of these data gives it refined meaning. Today, definitions of the term Market Access are as numerous as the people you ask…but everyone will use the word “value” in the definition, maybe not even mentioning the word “price.” You’ll notice, however, that those same people will show frustration with their definitions, because in the end, they’ll have to justify and validate a price…based on an ever-changing value story. Even the Academy of Managed Care Pharmacy (AMCP)—which established the industry standards for evaluating products for formulary placement, coverage, and reimbursement decisions in the US—says that “communications between manufacturers and healthcare decision makers should evolve over the product lifecycle as new evidence becomes available.” While launch-timed dossiers may rely to a greater extent on modeled projections based on clinical trial evidence and reasonable assumptions related to market dynamics and product uptake, new evidence describing the actual use and effect of the product in a real-world setting should be developed to inform formulary management across the product lifecycle. Ongoing generation of real-world evidence serves the important purpose of further defining and validating claims related to product value. (The AMCP Format for Formulary Submissions, Version 4.0; “Foreword”) The resulting definition of Market Access, then, is a dynamic process for building an ever-growing, ever-changing value story that validates a product’s price/position in healthcare practice. “An ever-growing and ever-changing value story” creates a continual challenge to keep the story up-to-date, to educate internal personnel continually, and to communicate with the market about complex real-world evidence…the scope and meaning of Market Access continues to evolve. |
Details
PurposeThis forum is for publishing and retaining articles on Medical Communications. If you have an ideas or would like to publish an article in this category please send a communication through the members page and we will review and either respond back or publish your article. Archives
April 2022
Sub Categories |
|
© COPYRIGHT 2018. ALL RIGHTS RESERVED.
|


 RSS Feed
RSS Feed
