By Steve Casey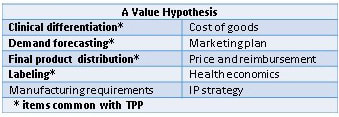 Early-stage companies commonly focus solely on investors and regulatory stakeholders and overlook other stakeholders who are critical in optimizing the later development-commercialization pathway. Addressing these other stakeholders maximizes the “downstream” value of any product. Such stakeholders are gatekeepers critical to life-cycle areas such as market access, reimbursement, and medical education. Even investors in early-stage companies are now considering the impact the “downstream” stakeholders have on their ability to secure financial exits. Today’s newly minted healthcare-product companies are frequently being asked to demonstrate both regulatory viability and commercial potential at the earliest stages. Long gone are the days when a product would “sell itself” and companies could count on guaranteed reimbursement and rapt attention from healthcare providers. Rather, today’s stakeholders are focused on specific therapies with defined risks and demonstrated healthcare benefit. As a result of these changes, a broader definition of product success must be established during early development to generate a rational basis and plan of action for both product approval and commercial success.
Engineer the Product Value It has long been understood that an evaluation of commercial potential during the early stages of clinical development in parallel with a well-developed regulatory strategy can greatly increase the value of a product. The integration of market forecasting, health economics, reimbursement and anticipated medical practice input leads to a development strategy that engineers more value and is more cohesive than a traditional staged approach. While commonplace in large pharmaceutical companies, this integration can be difficult for smaller companies where resources are limited, information is incomplete, and, frankly, product commercialization is viewed as the job of whatever company decides to license the product. While many legitimate limitations exist in the ability to describe a product’s potential completely at an early-development stage, future success will be determined by how well companies are able to structure integrated development programs to meet the needs of all stakeholders. How to Get There from Here Fortunately, it doesn’t require armies of healthcare professionals, scientists, and MBAs to develop an integrated clinical, regulatory, and commercial development plan. If a company takes a structured approach to establishing the program goals, much of the information is common to all perspectives. It has been said that the value of a product is driven by its product label; this is true if the commercial aspects of a healthcare product’s business are limited to communicating only what is in the label. To help guide development programs the FDA has provided a good starting point for drug developers with the Target Product Profile (TPP). The FDA TPP template follows the major categories of a package insert, allowing developers to forecast the desired target product attributes and track progress against this profile. The TPP tool is well understood and should always be used by companies developing new therapies. However, the TPP is focused primarily on regulatory approval and is not sufficient to describe fully the commercial attributes of a successful therapeutic. In parallel to the development of the TPP, companies should also consider creating a value hypothesis. The Value Hypothesis A value hypothesis considers the desired global commercial attributes for the therapeutic. As with the TPP, for early-stage programs the value hypothesis will be more general and aspirational in nature, but the real value of the hypothesis for an early-stage development program is in its creation. The development of the hypothesis, the identification of unknowns, and the specific questions that are generated as a result of the analysis, will all guide where early-stage candidates may warrant additional commercialization diligence. While all items will be important eventually, prioritizing where a company should focus limited resources in the value hypothesis is an important decision. A Value Hypothesis The importance of intellectual property (IP) protection in the biopharma industry dictates that IP strategy should be the top priority. Although many companies have competent legal staff handling their IP strategy, many early-stage programs stop with IP and provide limited focus to the other important elements that define commercial value of an early-stage therapeutic (ie. clinical differentiation, pricing/reimbursement, and demand forecasting). An exhaustive analysis of these commercial components is not practical or necessary at the early stages of product development; however, any company seeking to invest $10M or more in getting to a Phase 1 clinical study should have a clear understanding of the commercial assumptions for their product. But a value-hypothesis analysis is extremely valuable and better informs clinical decisions. Starting with a value hypothesis, therapeutic developers will improve clarity of the development/commercialization pathway, enhance confidence in the clinical program, and create a solid understanding to address stakeholder needs. This integration of regulatory and commercial factors will be critically important for product success in the new healthcare-market dynamics. The senior staff at Omni Healthcare Communications have extensive expertise in creating value hypotheses for developmental agents during their experiences while working at pharma companies and as proactive agency partners. Omni welcomes the opportunity to bring an unparalleled depth and breadth of experience to maximize the benefit and value that your product can achieve.
0 Comments
by Michael Caso  Biopharmaceutical companies pride themselves on conducting comprehensive research activities. These could be clinical programs to determine the viability of a new chemical entity to satisfy an unmet medical need, market access programs, or more recently, patient-centric needs of targeted populations. Why then have these same companies virtually ignored the medical, social and cultural needs of the fastest growing segment of the U.S. population? Ethnic and racial minorities currently comprise over 30 percent of the U.S. population and will grow to 50 percent by 2050. In fact, in many geographical areas, minority populations represent an “emergent majority” — a collective majority of minorities. Additionally, this population can have unique responses to medications and are differentially impacted by a number of common disease states for which they are diagnosed later and have poorer control or survival rates than majority populations. It would appear that there exists a nexus of needs and opportunities that should appeal to the biopharmaceutical industry, where a focus on a medical need can result in a healthier population and a healthier bottom line. This is the first of a three-part posting that will provide supportive data that identifies the need, demonstrates targeting opportunities, and provides specific strategies and tactics designed to positively impact complementary health disparity reduction and business goals. |
Details
PurposeThis forum is for publishing and retaining articles on Medical Communications. If you have an ideas or would like to publish an article in this category please send a communication through the members page and we will review and either respond back or publish your article. Archives
April 2022
Sub Categories |
|
© COPYRIGHT 2018. ALL RIGHTS RESERVED.
|

 RSS Feed
RSS Feed
